Evidence-based Medicine: What you need to know
Evidence-Based Medicine (EBM) was started by a group of healthcare professionals at McMaster University, in Hamilton, Canada. This group thought that patients had the right to know the evidence about the benefits and risks when making decisions about their health care, including what treatments may be best for them. Instead of the antiquated sentiment, “Trust me, I’m a Doctor,” this decision-making process focuses on empowering the patient to make more informed choices in their healthcare.
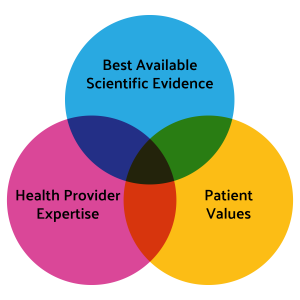
Evidence-based medicine is a careful approach that aims to strike a balance between 3 essential components: the best available scientific evidence, the patient’s values and the expertise of the health provider.
There is a lot of information out there about how to maintain your mental health and how to treat a mental illness. It can be hard to know what information you can trust!
When it comes to gathering and compiling scientific evidence, there are two broad types of research: quantitative and qualitative. Both approaches are valuable! Depending on the research question and other factors, either one or a combination of the two, may work best.
Quantitative research is numerical in nature. The data is analyzed by using mathematical and statistical techniques. Quantitative research can test a hypothesis and is well-suited to collecting information from larger groups or samples. As such, the results can often be generalized to wider populations. Surveys and scales are commonly used tools in quantitative research.
Qualitative research, on the other hand, does not use numerical data, but rather focuses on finding patterns and themes. It is a descriptive process, drawing on words, stories and experiences. Qualitative research aims to capture an in-depth understanding of how or why something has happened. Interviews and focus groups are common practices in qualitative research.
Unfortunately, not all information you find – for example, on the internet, in books or in magazines – is equally correct. That’s where understanding the Levels of Evidence can help.
Evidence is Hierarchical: Levels of Evidence
Health professionals and scientists carefully follow a rigorous process (ex. research), to determine if a health care treatment works, who it will work best for, and if it is safe. Scientific evidence is ranked hierarchically, based on the strength of the results obtained. This is called Levels of Evidence. The Levels of Evidence can help us consider how robust or how trustworthy the information is about a given health or mental health intervention.
Levels of Evidence*
Levels of Evidence*
A systematic review is basically a study of studies. It attempts to collect all the existing evidence on a certain topic in order to answer a specific question. It uses a predefined methodology deciding what information will be included and excluded before starting the literature search. Systematic reviews often include meta-analysis. Such reviews, when conducted on high quality RCTs, are positioned at the highest level in the hierarchy of clinical evidence.
A meta-analysis (meaning, an analysis of analyses) is a statistical method that pools, or combines the samples of several similar studies, typically to determine if an intervention is effective, and the effect size. The reason for doing this is that the bigger the sample size is, the more power scientists have to determine whether an intervention is effective, and how much impact it has.
Randomized Controlled Trials (RCTS) are typically used to compare two or more treatments, in which participants are randomly assigned to a treatment or control group. The investigators and the participants are ‘blinded’ meaning they are unaware of who is getting what treatment. This ensures that their observations are not influenced (biased). This type of study is good to establish the effectiveness of treatments. Despite RCT being considered one of the highest levels of evidence, not all RCTs are conducted the same way or with the highest standards. Therefore, the results of any studies need to be carefully analyzed.
Quasi-Experimental Designs are typically used in certain situations, such as community or field research settings, when a classic RCT is not feasible. This is when a quasi-experimental design can be useful. The prefix quasi means “resembling or almost.” Quasi-experimental research involves the manipulation of an independent variable without the random assignment of participants to conditions. Notable types of quasi-experimental research include non-equivalent group designs, pre-test/post-test, and interrupted time-series designs.
Cohort studies are sometimes called a longitudinal design because a cohort, or group of patients (study participants) who share certain characteristics (ex. risk factor, diagnosis, involvement in a treatment intervention) are followed across time. This method includes studies that follow and compare cohorts in a prospective manner (looking forward) or retrospective manner (looking backward).
Prospective Comparative studies are when researchers follow the participants for a certain period of time to see if they developed a particular disease. Then, the group is compared with a similar group of people who have not developed any health condition. Prospective studies can help to determine risk factors.
Retrospective Comparative Studies are when researchers look back through existing data sets. For example, a researcher could study two groups of people that share some similarities. However, one group has developed a disease and the other has not. The researcher looks back into their past medical history to see which factors could be associated with a disease or health problem. Retrospective comparative studies are commonly used in health and psychological research.
Cross-Sectional Studies are observational in nature and involve looking at data from a population at one specific point in time. The participants in this type of study are selected based on particular variables of interest. The benefit of a cross-sectional study design is that it allows researchers to compare many different variables at the same time.
Case Series are a description of several cases, and their relationship to one another. For example, the investigator will report on four different cases of patients who had an unusual response to a specific therapy or treatment. Limitations include a small sample size (therefore not generalizable results), and they don’t test a specified hypothesis.
Case Reports are a detailed description of a clinical case including the diagnosis, treatment and response to treatment of one patient. For example, the researcher might write about a rare disease or an unexpected side effect to a particular treatment.
Anecdotal Evidence means that people believe something works, and stories are told and re-told about something working, but it hasn’t been tested to see if that thing actually does work. This level of evidence does not come from research and does not follow the steps of the scientific process. Rather, it is based on someone’s opinion or belief. A lot of information you will find about health care, especially from the internet is anecdotal and does not come from research.
* This Figure represents the classic hierarchy of evidence-based medicine, in which the highest level of evidence is thought to be derived from RCTs (or systematic reviews and meta-analyses based on RCTs). However, for certain types of research questions, RCTs are neither possible nor desirable and other types of designs may provide the best possible evidence. For example, in Implementation Science (focused on maximizing the adoption, appropriate use, and sustainability of effective clinical practices in real world settings), “quasi-experimental designs” are deemed to be more appropriate for certain questions (Miller CJ, Smith SN, Pugatch M. Experimental and quasi-experimental designs in implementation research. Psychiatry Res. 2020 Jan;283:112452. Epub 2019 Jun 20. PMID: 31255320).
How to Evaluate Research: Some Questions to Ask
What is the study design? (ex. Is it a case study? An RCT?) Where does it fit on the evidence hierarchy?


